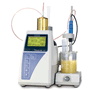
Kjeldahl
Product selection |
|
Processing your request... |
|
Processing your request... |
 Chemicals One of the largest selection of chemicals from one source...
Chemicals One of the largest selection of chemicals from one source...
 VWR® anti-vibration slab and weighing table Polished granite anti-vibration slab. Four absorption feet enhance...
VWR® anti-vibration slab and weighing table Polished granite anti-vibration slab. Four absorption feet enhance...
 VWR® traceable weight sets, class F1, with calibration certificate Anti-magnetic, polished stainless steel, wire weights up to 500 mg...
VWR® traceable weight sets, class F1, with calibration certificate Anti-magnetic, polished stainless steel, wire weights up to 500 mg...
 Chemicals One of the largest selection of chemicals from one source...
Chemicals One of the largest selection of chemicals from one source...
 e-Commerce / VWR Mobile Technology Solutions Small unit with powerful shaking action.
e-Commerce / VWR Mobile Technology Solutions Small unit with powerful shaking action.
 Chemicals One of the largest selection of chemicals from one source...
Chemicals One of the largest selection of chemicals from one source...
 e-Commerce / VWR Mobile Technology Solutions Small unit with powerful shaking action.
e-Commerce / VWR Mobile Technology Solutions Small unit with powerful shaking action.
 Asecos Safety Cabinets European leader in the storage solution for chemicals, asecos offers a wide range of safety cabinets to protect your property and your persons.
Asecos Safety Cabinets European leader in the storage solution for chemicals, asecos offers a wide range of safety cabinets to protect your property and your persons.
 e-Commerce / VWR Mobile Technology Solutions Small unit with powerful shaking action.
e-Commerce / VWR Mobile Technology Solutions Small unit with powerful shaking action.
 Asecos Safety Cabinets European leader in the storage solution for chemicals, asecos offers a wide range of safety cabinets to protect your property and your persons.
Asecos Safety Cabinets European leader in the storage solution for chemicals, asecos offers a wide range of safety cabinets to protect your property and your persons.
 e-Commerce / VWR Mobile Technology Solutions Small unit with powerful shaking action.
e-Commerce / VWR Mobile Technology Solutions Small unit with powerful shaking action.
![]()
Avantor Services provides a wide range of specialized services and digital solutions to help you solve complex challenges.
We’ve built our reputation on consistent, comprehensive mastery of day-to-day operations, allowing lab, clinical, and production environments to focus their high-value resources on core scientific priorities.
As our customers’ needs have evolved, so have our capabilities. We have become experts in scientific operations, improving performance with sophisticated solutions and providing guidance on best practices.
You can select and customize services for peak efficiency, quality, and accelerated innovation.
Contact us at services@avantorsciences.com.
 e-Commerce / VWR Mobile Technology Solutions Small unit with powerful shaking action.
e-Commerce / VWR Mobile Technology Solutions Small unit with powerful shaking action.
 Avantor Services Providing a wide range of specialized services and digital solutions to help you solve complex challenges.
Avantor Services Providing a wide range of specialized services and digital solutions to help you solve complex challenges.
 e-Commerce / VWR Mobile Technology Solutions Small unit with powerful shaking action.
e-Commerce / VWR Mobile Technology Solutions Small unit with powerful shaking action.
|
{0} Item(s) added to your Shopping Basket
|
||||||||
|---|---|---|---|---|---|---|---|---|
|
||||||||
Due to maintenance activity, Global web site will not be available from 6AM till 1 PM ET on 24th August 2024

|
|
|||||
|
Avantor® is a leading life science tools company and global provider of mission-critical products and services to the life sciences and advanced technology industries. We work side-by-side with customers at every step of the scientific journey to enable breakthroughs in medicine, healthcare, and technology. Our portfolio is used in virtually every stage of the most important research, development and production activities at more than 300,000 customer locations in 180 countries. For more information, visit avantorsciences.com and find us on LinkedIn, X (Twitter) and Facebook .
© 2024 VWR International, LLC. All rights reserved..